HFEA’s written evidence for WEC inquiry into egg donation and freezing
Please see the HFEA’s written evidence submitted as part of the Women and Equalities Committee (WEC) inquiry into egg donation and freezing. This evidence can also be found alongside other submissions on the UK Parliament website.
Table of contents
- The HFEA
- Summary
- What are the short and long-term health impacts of donating or freezing eggs and embryos and to what extent are they sufficiently researched and understood?
- Whether the counselling provided ahead of egg donation is adequate to ensure informed consent, including of potential health impacts?
- What level of compensation / payment should be provided to egg donors, if any?
- What evidence is there, if any, of vulnerable women being encouraged into egg donation or egg freezing?
- Is the regulatory regime on advertising as it applies to egg donation and people wishing to freeze their eggs or embryos sufficient?
- What has been the impact of changes to the release of donor information, including support for the families involved, and whether further legislation is required?
- Whether the existing legislative framework, including the Human Fertilisation and Embryology Act 1990, is effective in safeguarding the best interests of those undergoing treatment for egg donation or egg or embryo freezing?
- Annex: HFEA data on egg donation and freezing
- References
The HFEA
The Human Fertilisation and Embryology Act 1990 (HFE Act) established the Human Fertilisation and Embryology Authority (HFEA) and the regulatory regime which we oversee. As the independent regulator of fertility treatment and human embryo research in the UK the HFEA aims to ensure that everyone who steps into a licensed fertility clinic receives high quality care. We do this by licensing, inspecting and setting standards. We also provide free, clear and impartial information about fertility treatment, clinics, and egg, sperm, and embryo donation.
All clinics are inspected at least every two years and must demonstrate that they comply with the relevant law, licence conditions and HFEA Code of Practice. Where we have concerns, inspections will be more frequent and a clinic monitored closely through enhanced regulatory oversight such as more frequent contact, onsite visits or unannounced inspections. It is a legal requirement to report incidents and it would be a serious matter if any incidents were uncovered on inspection which hadn’t been reported. The HFEA also collects patient feedback from every clinic which is reviewed in advance of inspection and any concerns are discussed with the clinic who must set out how these concerns are being addressed.
The HFEA holds one of the longest running databases of fertility treatments and outcomes in the world, going back to 1991 and we publish this data, showing trends in treatment types, demographics and outcomes on our website and in regular reports.
Our sponsor is the Department of Health and Social Care and we work closely with several professional and patient bodies, charities and organisations, as well as our patient engagement forum with recent or current patients, partners, surrogates, intended parents, donor-conceived people over the age of 18 and parents of donor-conceived people.
We publish all our governance and licensing papers and minutes and our Authority (Board) meetings can be attended by anyone, or watched online afterwards.
We have a statutory duty to publish information relating to fertility treatments and provide free and impartial information on our website and through our social media channels as well as presenting at patient and professional facing events.
Summary
The HFEA has provided information relating to each aspect of the Committee’s Terms of Reference based on our regulatory role and data.
Where we have concerns about any of the areas relating to this Inquiry, we have briefly set these out and hope to discuss this further in any oral evidence session.
Those who donate or freeze their eggs in UK licensed clinics do so within a tightly regulated system with strict rules set out in law and in the HFEA Code of Practice.
There is little direct evidence of short or long term health impacts of donating or freezing eggs and embryos. However, there is robust evidence on the impact of IVF more generally and there is little reason to believe that those donating or freezing eggs would be differently affected.
The HFEA does not hold data that suggests vulnerable women being encouraged to donate or freeze their eggs.
There is strong information provision set out in the law in advance of egg donation to ensure informed consent. However, the law as it stands only requires the offer of counselling and we believe this should be strengthened.
The UK system is based on altruistic donation. This is supported by a fixed level of compensation, set by the HFEA, to cover donor expenses and ensure there aren’t any unjustifiable barriers to donation. We do not have evidence that the UK compensation limits are driving donor behaviour.
The HFEA does not have powers over advertising but is concerned that some adverts do not portray the seriousness of egg donation and freezing or fertility treatment more broadly.
Fertility treatment with donors is increasing year on year and those involved may require different types of support at different times. The release or discovery of donor information can have varying effects on those involved. New legislation will be required to address the greater availability of genetic information through direct-to-consumer DNA tests or social media.
Aspects of the legislation are out of date and the HFEA made recommendations for change in 2023 on a wide range of areas outlined below.
At the end of our written evidence, we have provided an annex with data from the HFEA Register relevant to this inquiry.
What are the short and long-term health impacts of donating or freezing eggs and embryos and to what extent are they sufficiently researched and understood?
Summary
- There is little direct evidence of short or long term health impacts of donating or freezing eggs and embryos. However there is robust evidence on the health impact of IVF more generally and there is little reason to believe that those donating or freezing eggs would be differently affected.
- The process of freezing or donating eggs involves part of the IVF process, which is generally considered a safe medical treatment in the short and long-term.
- As a medical procedure, there are some risks, as well as possible longer term psychological considerations of both freezing eggs and being a donor.
- In 2024/25, there were more than 100,000 cycles of fertility treatment (including donation and freezing cycles) in UK licensed fertility clinics, with incidents occurring in less than 1% of cycles.1
- The HFEA website has patient information on the risks of fertility treatment, which was updated in 2024 following the most recent expert scientific review of the latest evidence.
Legislation and guidance
- There are strict requirements in HFEA Licence Conditions and the Code of Practice relating to the information that must be given before egg retrieval takes place in UK licensed fertility clinics, whether for the patient’s own use or to donate to others. This includes information about the potential immediate or longer-term health risks and the psychological consequences of being a donor, as well as the requirement to offer counselling to everyone involved.
- Donors must be selected on the basis of their age, health and medical history, provided on a questionnaire and through a personal interview performed by a qualified and trained healthcare professional. This assessment is to identify and screen out potential donors whose donations could present a health risk to others, such as the possibility of transmitting diseases, or health risks to themselves, including superovulation, sedation or the risks associated with the egg collection procedure or the psychological consequences of being a donor.
- Gametes for the treatment of others should not be taken from anyone under the age of 18.
- A prospective donor should not be accepted if the clinic concludes that a recipient or any child born as a result of treatment using the donor’s gametes is likely to experience serious physical, psychological or medical harm, or if the clinic cannot get enough further information to conclude there is no significant risk.
- We provide information about the risks of fertility treatment on our website for patients (including those freezing their eggs) and for egg donors.
Health impacts of the IVF process when donating or freezing eggs and embryos
Donating eggs or embryos involves part of the IVF process, including stimulating the ovaries and egg collection. Egg freezing requires the patient to undergo this same part of the process, with those wishing to later use their eggs in treatment continuing to embryo transfer and pregnancy, if the treatment is successful.
There are few studies looking specifically at the short or long-term health impacts of donating or freezing eggs or embryos, but we can look to the large number of robust studies assessing the short and long-term health impacts of undergoing IVF more generally as a guide.
Fertility treatments in the UK are generally very safe – most women are no more likely to experience problems with their health or pregnancy than women who have conceived naturally. However, there are some risks, which range from mild discomfort to more serious conditions. Short-term health risks of IVF include:
- Infection, bleeding, and bowel perforation following IVF or IUI procedures (rare).
- Ovarian Hyperstimulation Syndrome (OHSS), which is a reaction to fertility drugs taken to increase egg production. Around a third of women will have mild OHSS. Clinics must report any severe and critical occurrences of OHSS to us as an adverse incident. Our most recent data shows that in 2024/25, there were 67 cases of severe and critical OHSS reported by UK clinics.
Risks during pregnancy following fertility treatment include:
- having a multiple pregnancy or birth (twins, triplets or more)
- having an ectopic pregnancy
- a slightly increased risk of pre-eclampsia, gestational diabetes (diabetes occurring only during pregnancy), and high blood pressure (hypertension) compared to natural conception (these complications are rare, and research is still ongoing).
- some studies also found a slightly increased risk of post-partum haemorrhage (PPH) in patients who conceived following fertility treatment.
- some research has suggested that patients who undergo a programmed (medicated) cycle compared to a natural cycle frozen embryo transfer may be at increased risk of experiencing high blood pressure during pregnancy.
- possible birth defects (these are rare and research is still ongoing).
Complications during pregnancy can sometimes be linked to the health of the patient before getting pregnant, known as pre-conception health. For example, those with pre-existing cardiovascular risk factor conditions, such as obesity, high blood pressure (hypertension), or diabetes, are at higher risk of complications during pregnancy.
Risks associated with using donated eggs, sperm, or embryos, or having reciprocal IVF:
- Patients having fertility treatment using donated eggs or embryos have a slightly higher risk of complications during pregnancy, including pre-eclampsia and high blood pressure (hypertension), than those using their own eggs. These risks also apply to couples having reciprocal IVF, where the partner’s egg is used. There is no difference in the risk of pre-eclampsia during pregnancy for women who use donated sperm compared to those who use their partner’s sperm.
Scientific review of evidence on health outcomes for assisted reproduction patients
The HFEA Scientific and Clinical Advances Advisory Committee (SCAAC) last considered the evidence on health outcomes for assisted reproduction patients, including egg donors and gestational surrogates, in June 2025. The papers and minutes can be found on our website.2
Research considered by SCAAC included papers investigating the risk of obstetric outcomes following assisted reproduction treatments, such as pre-eclampsia and hypertensive disorders of pregnancy, the relationship between fertility drugs and cancer incidence later in life, as well as psychological issues.
The review found that for some topics considered, current evidence was limited or conflicting. For example:
- Evidence on breast cancer incidence in egg donors is inconclusive and conflicting, with no clear established link.
- Research investigating a link between the use of ovulation stimulating drugs and increased ovarian and thyroid cancer risk is conflicting.
- There is limited research on the long-term impact of cryopreservation on gametes and embryos, or whether health outcomes of assisted reproduction patients is related to the method used for embryo freezing.
In addition, the committee noted that findings from large-scale international studies will vary from the findings from UK based studies due to differences in the cultural and regulatory context.
Scientific review of evidence on health outcomes for egg donors
The SCAAC review of health outcomes for assisted reproduction patients also considered health outcomes for egg donors specifically.3 Studies reviewed reported on the short-term complications of the procedure, including OHSS and pain. They also evaluated psychological issues including anxiety amongst egg donors, and breast cancer incidence.
The review found that studies were conducted mostly via surveys completed by egg donors, rather than comparing complications experienced by egg donors to the general population, which would provide clearer evidence on health outcomes of donation specifically. In addition, several of the studies were focused on the health outcomes of egg donors based in the USA, where differences in the healthcare system and regulation of egg donation mean findings may not be relevant to the UK context. Additional large UK-based, long term, high quality studies would provide more robust data on the health outcomes for egg donors in the UK.
Following this evidence review we updated the patient information on our website to highlight new findings and update information about the role of preconception health in treatment outcomes.
Data on health impacts
The HFEA has collected data on all IVF and donor insemination treatments performed in the UK since 1 August 1991. We use this data to improve patient care and support researchers to conduct scientific research, whilst ensuring very strong protection of patient, donor and offspring confidentiality. You can read more about how HFEA Register data is used in research, and about ongoing and completed research projects, on our website.
Whether the counselling provided ahead of egg donation is adequate to ensure informed consent, including of potential health impacts?
Summary
- There is strong information provision set out in the law in advance of egg donation to ensure informed consent. However, the law as it stands only requires the offer of counselling and we should believe this should be strengthened. Please see section 7 for further details on HFEA recommendations for law reform in relation to this.
- By law, anyone having treatment in a UK licensed fertility clinic – including egg donation – must be offered counselling and provided with specific information before giving consent and starting treatment.
- Clinics must demonstrate that they comply with strict requirements set out in Licence Conditions and the HFEA Code of Practice on what and how information is given before egg retrieval takes place, whether for the patient’s own use or to donate to others. This includes information about the potential immediate or longer-term health risks to themselves, or the psychological consequences of being a donor, as well as offering counselling to everyone involved.
- All clinics are inspected by the HFEA at least every two years. At inspections, the clinic’s procedures are reviewed to ensure that they meet the requirements for providing information to patients and donors, and that consent is obtained before any licensed activity takes place. All inspection reports and decisions are published on the HFEA website.
Legislation and guidance
- Counselling and information provision are generally separate but overlapping processes. The HFE Act states that patients must be provided with relevant information and be offered counselling about the implications of their decision before consenting to treatment. Relevant information must be provided even if the donor does not wish to take up the offer of counselling.
- The provision of relevant information must include information about potential health risks, and the Code of Practice states that clinics should give patients enough time to consider the information before any treatment begins.
- Generally, the offer of counselling required by law would involve exploring the implications of the treatment and donation with a qualified counsellor.
- Information provision and counselling must be given by a trained professional and delivered in a way that the patient or donor will understand.
- HFEA inspections assess clinics adherence to the law, licence conditions and Code of Practice guidance, which by law must take place at least every two years.
HFEA assessment of counselling and information provision
The current framework for counselling provision and informed consent is detailed and the tight regulation of fertility clinics is considered a best practice model.
On inspection, clinics are required to demonstrate that they have offered counselling and information, provided by trained personnel in a way that the prospective patient or donor will understand and we check that clinics comply with these standards. We review records to ensure that information is provided, the offer of counselling is recorded, when the offer took place, that this was before the donor gave their consent, and whether the offer was accepted, and if so, when the counselling took place.
Recommendations from specially trained inspectors with relevant scientific or clinical backgrounds are then taken to a HFEA licence committee to assess whether a clinic licence can continue or be renewed. This acts as a robust assurance mechanism. All inspection reports including assessments of information provision and counselling are published for every UK licensed clinic on the HFEA website.
On a periodic basis, the HFEA carries out a national fertility patient survey to gather views of a representative sample of fertility patients across the UK who have had recent fertility treatment in licensed clinics. The most recent survey looking at all patients who had treatment as whole (including egg donors) showed that most patients found communication around consent forms was clear (93%).4 When looking at satisfaction with various aspects of treatment, overall satisfaction for all aspects of treatment was above 65%, with the highest satisfaction with the extent to which patients understood what they were consenting to (89%). The lower end of satisfaction (at 69%) was with level of support given, including access to counselling.
Information provision
The HFE Act requires that patients receive relevant information before consenting to treatment. This information must be provided even if the donor does not wish to take up the offer of counselling. HFEA Licence Conditions specify further details about information to be provided, including information about the nature of the treatment, its consequences and risks, relevant storage periods, the right to withdraw or vary their consent, and the availability of counselling.
Information about risks should include potential immediate and longer-term risks of the treatment and any treatment add-ons used. This includes the possibility of developing ovarian hyperstimulation syndrome (OHSS), information about the possible symptoms of OHSS, and what the woman being treated should do and who to contact if experiencing symptoms of OHSS. Information should also include the possibility of any children conceived having developmental and birth defects, the nature and potential risks of any alternative treatment options available so the patient can make an informed decision about their treatment, and the possibility of emotional distress.
Guidance Note 4 of the Code of Practice sets out further requirements for information provision, including about success rates, cost of treatment, and information that should be included in any contractual arrangements between patients and centres.
Counselling
The HFE Act states that counselling must be offered in certain circumstances including before egg donation or egg freezing. The HFEA Code of Practice (Guidance Note 3) sets out the expectations for counselling provision and patient support. This includes that clinics should:
- inform patients and donors about how to access counselling and give them sufficient time to consider the offer before beginning any treatment (3.2), that counselling about donating gametes and embryos requires separate attention to any more general counselling about treatment that that individual may be undergoing (3.4) and that counselling should be available before, during and after treatment – including if someone returns to the clinic after the treatment or donation process is completed (3.5).
- maintain a patient support policy “to outline how the centre ensures that patients, donors and their partners (where applicable) receive appropriate psychosocial support from all staff they encounter before, during and after treatment. Psychosocial support is delivered by all members of staff and includes, but is not limited to, access to counselling.” (3.14). We provide examples of Patient Support Policies on our Clinic Portal.
The Code of Practice states that clinics should have at least one dedicated counsellor who should hold a relevant, recognised qualification and who is accredited or working towards accreditation by the British Infertility Counselling Association (BICA) (or an equivalent specialist fertility counselling body) – see the Code of Practice (2.15).
Egg donation
The Code of Practice sets out the information that should be provided to prospective donors before any consents or samples are obtained. This is set out in full at 11.36, and includes information about the procedure (including any discomfort, pain and risks), reasons for and potential consequences of health screening and genetic testing, and the procedure for donors to withdraw consent for the use and storage of their gametes, or embryos created with their gametes. It also includes information on legal parenthood of any child born, what information about the donor must be collected by the clinic and held on the HFEA Register, and what information may be disclosed to prospective recipients or to the parents of donor-conceived children.
The Code of Practice also sets out additional requirements for information provision for women participating in egg sharing arrangements (Guidance Note 12). Before treatment begins, the clinic should give the gamete provider and the recipient written information, including on how the clinic proposes to distribute the gametes between the provider and the recipient(s), the terms of the agreement to be made, and the law relating to consent, in particular the rights of a person providing gametes to vary or withdraw consent, and the implications of doing so.
What level of compensation / payment should be provided to egg donors, if any?
Summary
- The UK system is based on altruistic donation. This is supported by a fixed level of compensation, set by the HFEA, to cover donor expenses and ensure there aren’t any unjustifiable barriers to donation. We do not have evidence that the UK compensation limits are driving donor behaviour.
- Payment for gametes (eggs and sperm) is not permitted in the UK. Providing or receiving unauthorised payment or benefit for supply of gametes or embryos is a criminal offence.
- Egg donors can receive compensation of up to £985 per donation ‘cycle’ to cover their costs. Sperm donors can receive up to £45 per clinic visit.
- The HFEA sets out strict requirements in its Directions, Licence Conditions and Code of Practice relating to the way that clinics can compensate donors.
- Donation has life-long consequences for everyone involved - the donor and their family, the recipient(s), and any children born – and the system for donor compensation aims to strike the balance between covering donor expenses and ensuring donors feel valued, whilst not removing the altruistic motivations behind donation.
Legislation and guidance
- The HFE Act sets out that payment of donors in the UK is not permitted and that no money or other benefit must be given or received in respect to any supply of gametes, embryos or human admixed embryos unless authorised by Directions.
- Providing or receiving unauthorised payment or benefit for supply of gametes or embryos is a criminal offence. Section 41(8) of the Act states: Where a person to whom a licence applies or the holder of the licence gives or receives any money or other benefit, not authorised by Directions, in respect of any supply of gametes, embryos or human admixed embryos, he is guilty of an offence.
- The HFEA sets out strict requirements its Licence Conditions and Code of Practice relating to the way that clinics can compensate donors, including that they must not accept an individual as a donor who is known (or is reasonably suspected) by that clinic to have received or to be about to receive money or other benefits not in line with HFEA Directions.
- Egg donors can receive compensation of up to £985 per donation ‘cycle’ to cover their costs (a donation cycle is one complete round of treatment, at the end of which the eggs are collected and donated). Sperm donors can receive up to £45 per clinic visit to cover their expenses,
- Clinics may compensate donors with an excess amount in cases where expenses (such as for travel, accommodation or childcare) exceed the amounts specified in HFEA Directions. Clinics may only provide excess expenses which: (a) are reasonable (b) do not include loss of earnings (c) have been incurred by the donor in connection with the donation of gametes provided to that clinic, and (d) have been incurred by the donor solely within the UK.
Compensation amount for egg and sperm donors in the UK
The compensation amount for egg and sperm donation is set by the HFEA and is based on a system of altruistic donation. Academic research in the UK suggests that donating eggs and sperm is driven by altruism to help others to create families.5
Donor compensation at a fixed level was introduced by the HFEA in 2011, following a thorough ethical review, which identified a set of principles that ensured altruism remained at the heart of donation and that there weren’t any unjustifiable barriers to donation. As noted above, donors could be compensated an excess amount in cases where expenses (such as for travel, accommodation or childcare) exceed these amounts as long as the expenses are reasonable, and in connection with their donation within the UK. Clinics can compensate donors who are not permanent residents of the UK in the same way as UK donors but are not permitted to directly or indirectly pay the overseas travel of a non-UK donor.
Over the last 10 years the number of UK egg and sperm donors have largely remained similar but the number of patients seeking treatment with donor eggs or sperm has almost doubled from around 5,200 in 2013 to 9,200 in 2023 (in particular the number of single patients and those in female same sex couples having treatment with donor sperm).
The HFEA increased the amount of compensation in 2024 to take account of inflation and cost of living since the last change in 2011. The amount increased from £750 to £985 for egg donors (per donation cycle) and £35 to £45 for sperm donors (per clinic visit). The impact of inflation, particularly since 2020-21, meant that in 2024 the donor compensation rates were not reflective of the intended monetary value when the rates were set in 2011.This was the first time donor compensation had been reviewed since 2011.
HFEA inspections
On inspection, clinics would need to demonstrate that they have adhered to law, standards and guidance relating to donor compensation. This is reviewed and any concerns would need to be addressed by the clinic.
If the person responsible or the licence holder gives or receives any money or benefit for the supply of gametes, embryos or human admixed embryos that is not authorised by the applicable HFEA Directions, they have committed a criminal offence. Conviction may result in a prison term, a fine or both.
International comparisons
The position in respect of financial compensation or payment varies considerable across the world. The HFEA is aware that in some countries there are large financial benefits from being a donor, while in others compensation is not allowed and in treatment with donation is prohibited. The data from the UK shows that the number of egg and sperm donors has remained similar over many years which suggests that the amount of compensation amount is not the driving factor for donor behaviour. However, we are aware of concerns that have been raised over some advertising where donation is portrayed in a less serious way then we would want it represented.
What evidence is there, if any, of vulnerable women being encouraged into egg donation or egg freezing?
Summary
- The HFEA does not hold data that suggests vulnerable women being encouraged to donate or freeze their eggs.
- Those who donate or freeze their eggs do so within a tightly regulated system with strict rules set out in law and in the HFEA Code of Practice.
- The numbers of women donating eggs in the UK has remained similar in recent years6 and the median age of donors undergoing cycles to collect eggs for the primary reason of donation has remained steady over the last ten years or so.7
- Between 2011-2020, egg donors lived in comparable areas to the general population.8
- There are strict requirements set out in relation to the recruitment of donors and information that must be given to egg donors in advance of donating includes information about the potential immediate or longer-term health risks and the psychological consequences of being a donor, as well as offering counselling to everyone involved (see our response to Questions 1 and 2 above).
- Research suggests a range of motivations for freezing. Some freeze for medical reasons but the large increase in recent years has been for ‘elective’ egg freezing.
- Egg freezing should not be presented as a guarantee to having a baby in the future, and we expect clinics to follow consumer law and advertising guidance for UK fertility clinics produced by the CMA and ASA.
- We are concerned about the impact on potentially vulnerable women of the growing popularity of apps, websites and social media sites which encourage women to use a sperm donor outside a licensed clinic.
- Please see the Annex for further HFEA data on patients who donate or freeze their eggs.
Suitability of egg donors
Those who donate or freeze their eggs do so within a tightly regulated system with strict rules set out in law and in the HFEA Code of Practice. We have seen no evidence of vulnerable women being encouraged to donate or freeze their eggs.
Clinics are required to consider the suitability of the donor. If the clinic feels the donor is unsuitable then they should not be accepted.
As noted earlier HFEA licence condition T52 and Code of Practice Guidance Note 11 state that donors must be selected on the basis of their age, health and medical history, provided on a questionnaire and through a personal interview performed by a qualified and trained healthcare professional. This assessment is to identify and screen out potential donors whose donations could present a health risk to others, such as the possibility of transmitting diseases, or health risks to themselves, including superovulation, sedation or the risks associated with the egg collection procedure or the psychological consequences of being a donor.
Code of Practice guidance specifies ages of prospective donors, including that gametes for the treatment of others should not be taken from anyone under the age of 18.
Please see our response to Question 2 for further information on counselling and informed consent, and Question 1 on the requirements regarding the assessment of prospective donors.
Demographics of patients donating their eggs
The median age of donors undergoing cycles to collect eggs for the primary reason of donation in 2019-2023 was 30, compared to a median age of 31 in 2009-2013.9
Our Trends in egg, sperm and embryo donation 2020 report, published in 2022, found that egg donors lived in comparable areas to the general population.
Egg donations used in fertility treatment in the UK are typically from UK donors, with only around 3% of donor eggs imported from abroad. One reason that patients may import donor sperm or eggs is to find a donor of matching ethnicity.10One reason that patients may import donor sperm or eggs is to find a donor of matching ethnicity.
Demographics of patients freezing their eggs
The average age of patients freezing their eggs was 35 and the average age of thawing was 40 in 2023.11
The HFEA does not collect data on the socioeconomic background of patients who freeze their eggs. Research is limited but suggests women undergoing elective egg freezing may be more likely to be from higher socioeconomic groups and have high levels of education.12
Egg freezing data collected by the HFEA does not distinguish between reasons for freezing eggs, such as elective fertility preservation or for medical reasons. However, using NHS funding as a proxy for medical egg freezing indicates that at least 16% of egg freezing cycles were for medical reasons in 2018-22.13
Research on this topic is limited, but suggests that there are a range of reasons why patients may decide to freeze their eggs for non-medical reasons. The absence of the right partner with whom to start a family is often identified as a primary reason, alongside wanting to feel financially secure or ready for parenthood, and concerns about fertility declining with age.14 Social, cultural and religious approaches to motherhood and to assisted reproductive technologies have also been found to shape women’s perception of the procedure.15
Sperm donation outside of licensed clinics
We would like to draw the Committee’s attention to the related issue of apps, websites and social media sites which encourage people to use a sperm donor outside a licensed clinic. This may expose women to serious medical, legal and emotional risks.
A recent court case and media coverage of prolific private donors has highlighted some of these concerns, with judges warning about unregulated sperm donation and specific donors operating through social media.16 New services entering the market, an increase in groups accessing treatment who are largely ineligible for funding, and the general decrease in NHS funding for fertility treatments, suggest that unlicensed donation is becoming more prevalent.
It is always safer to have treatment with donor sperm at a HFEA licenced clinic, where there are laws and guidance to protect and support patients and donors. Clinics are required by law to ensure that donors, patients, and any future children are protected by carrying out rigorous health tests, ensuring legal parenthood consents are in place, and offering everyone involved counselling to ensure they are completely informed and comfortable with their decision.
Is the regulatory regime on advertising as it applies to egg donation and people wishing to freeze their eggs or embryos sufficient?
Summary
- The HFEA does not have powers over advertising but is concerned that some adverts do not portray the seriousness of egg donation and freezing or fertility treatment more broadly.
- Regulation of adverts is under the remit of the Advertising Standards Authority.
- The HFEA powers are limited in this area and do not generally allow us to tackle the commercial aspects of the modern fertility market.
- Any adverts for egg donation or egg freezing should be carried out in line with the HFEA Code of Practice, advertising codes of practice and consumer law.
Legislation and guidance
- Advertising of fertility treatment, including egg donation and egg and embryo freezing, is regulated by the Advertising Standards Authority (ASA) under the UK advertising codes and consumer law.
- The HFEA role is limited to setting standards through licence conditions and Code of Practice.
- Advertising and marketing for fertility treatment, including egg donation and egg freezing, should be carried out in a sensitive way reflecting the complex issues, medical procedures, any risks and long-term implications of treatment (13.1 of the HFEA Code of Practice)
- The Code of Practice sets out that adverts aimed at recruiting egg donors should not refer to the possibility of financial gain, or similar advantage (although it may refer to compensation permitted under HFEA Directions (11.1 of the HFEA Code of Practice).
Joint working with advertising and consumer law regulators
Negotiating the modern fertility sector as a patient can be challenging, not least because of the high emotional and financial stakes involved.
As the fertility regulator, we have long been concerned about how some clinics offer their services, but our regulatory powers do not allow us to tackle most of the commercial aspects of the modern fertility market.
The HFEA has therefore worked with the Competition and Markets Authority (CMA) and the ASA to develop guidance for fertility clinics and a HFEA/ASA enforcement notice for the fertility sector, setting out what information clinics should provide to patients, and when, to support informed decision-making.
Looking at adverts for fertility clinics and treatment in general, the Committee of Advertising Practice (CAP) published a report in December 2025 that found high levels of compliance in the fertility sector. It found that 96.7% of the ads assessed did follow the advertising rules. In the small number of cases where advertisements weren't compliant, effective action was taken, with clinics being contacted and instructed to amend or remove problematic claims from their paid advertising. Clinics have responded positively to contact, amending or removing such claims. One clinic was referred to the platform hosting its advertisement for enforcement.
We are pleased that the CAP will continue to work closely with clinics to address any remaining issues, ensuring that a high level of compliance across the sector is maintained and those considering fertility treatment continue to be protected from misleading advertising.
What has been the impact of changes to the release of donor information, including support for the families involved, and whether further legislation is required?
Summary
- Fertility treatment with donors is increasing year on year and those involved may require different types of support at different times. The release or discovery of donor information can have varying effects on those involved. New legislation will be required to address the greater availability of genetic information through direct-to-consumer DNA tests or social media.
- The HFEA is required by law to maintain a register of all licensed treatments, including those involving donation.
- Before 2005, donation was anonymous. In 2005 the law changed to allow a person conceived using a donor registered after 1 April 2005, to apply for identifiable donor information from the age of 18. This has given donor conceived people, providing they have been told they are donor conceived, an opportunity to learn about where they came from. The HFEA also gave donors non-statutory access rights to anonymous information about their genetic offspring, including the number, sex, and year of birth of people born as a result of their donation (now a statutory right following the 2008 amendments to the HFE Act).
- In October 2023, the first cohort of donor-conceived people were able to apply to the HFEA for identifiable donor information. This is known as an ‘Opening the Register’ (OTR) request.
- By the end of 2024, around 766 donor conceived people were eligible to request identifying information about their donor from the HFEA and by 2030, this will rise to around 11,427.
- To support this transition, in 2023 we launched the #WhoIsMyDonor campaign, to highlight the information available to people affected by egg and sperm donation and how they can apply for it. We improved our website content and signposting relating to donation, introduced new systems to increase our effectiveness in processing applications and recruited additional staff to manage the expected increase in requests for information. We will continue to monitor our OTR service as requests for information rise.
- The HFEA made a range of proposals on changes to the HFE Act to improve the position for donors, donor-conceived people and their families, including on access to donor information. See section 7 for further detail on this.
Legislation and guidance
- The HFE Act requires the HFEA to keep a Register of information about donors and treatments involving the use of donor gametes and embryos in the UK since August 1991. The Register also records the notified births resulting from these treatments.
- The Human Fertilisation and Embryology Authority (Disclosure of Donor Information) Regulations 2004 set out that from 1st April 2005 people were no longer able to donate sperm, eggs or embryos anonymously. Before this change donors were automatically anonymous (although a small number of donors have since chosen to remove their anonymity).
- This change allows most people conceived from donations made after 1st April 2005 to request identifying information from the HFEA about their donor after they turn 18. This includes the donor’s full name, last known address, date of birth and place of birth.
- Donors can find out the number, sex and year of birth of any children born from their donation(s).
- Applications for Register information can be made by donor-conceived individuals, donors and parents. Information can be requested from the HFEA via the Opening the Register service.
- The law requires clinics to offer counselling to donors and patients prior to donating or having treatment with donor eggs, sperm or embryos. HFEA Code of Practice guidance states that donors who are identifiable should be able to access counselling via the clinic at which they donated (see also response to Question 2).
- Parents of donor-conceived people can also contact the clinic they had treatment at for support. Support for those affected by donation can also be found from organisations such as the Donor Conception Network and Donor Conceived UK. The HFEA provides information and signposting on our website:
Support for donors and donor-conceived people
The HFEA is aware of the impact the disclosure of donor information can have on donors, donor-conceived people and their families, whether this information is accessed through the HFEA, or through the use of DNA testing and online matching services.
We provide detailed information on the HFEA website about the different kinds of support and counselling donors, donor-conceived people and their families may wish to access, along with links to organisations providing support: Counselling and support for donor-conceived people and donors.
Donor-conceived people and their families will want to access information about their donors at different times, with some wanting to try and find out about their child’s donor – by using DNA testing – when their child is young and others leaving it to the child to apply to the HFEA for information (should they want to) once they are 18. For those donor-conceived people who are eligible to get identifiable information about their donor, some are ready to do so as soon as they can, while for others, it is less important until they are older, or perhaps considering having children themselves. Organisations and counsellors can help individuals and families think through these potentially difficult issues.
Whether the existing legislative framework, including the Human Fertilisation and Embryology Act 1990, is effective in safeguarding the best interests of those undergoing treatment for egg donation or egg or embryo freezing?
Summary
- Aspects of the legislation are out of date and the HFEA made recommendation for change in 2023 on a wide range of areas outlined below.
- The HFE Act established the HFEA and the regulatory regime which we oversee.
- Much of the original 1990 Act remains fit for purpose and provides an effective framework for the regulation of a fast-developing area of medicine and research.
- However, the HFEA has recommended 14 changes to the Act (five of which are relevant to the Committee’s inquiry) to keep pace with significant changes in the size and structure of the fertility sector, in popular attitudes to fertility treatment, challenges to the statutory information relating to donor treatment, and in clinical and scientific developments. Many UK regulators have a wider and more effective range of powers to improve compliance and protect patients and consumers than those available to the HFEA.
- Any changes to the Act are decisions for the UK Government and ultimately Parliament.
Recommendations for legislative change with relevance to safeguarding the best interests of egg donors and patients freezing their eggs or embryos include:
- That the HFE Act should be revised to include an over-arching focus on patient protection. It is striking that there is no patient protection principle in the Act. Unusually in UK healthcare the majority of patients pay for treatment themselves, and so this consideration is not only about medical care and treatment but also about patients as consumers.
- That the Act should be revised to accommodate developments in the way fertility services are provided. More fertility services are being offered that fall outside the remit of the Act. Bringing all related UK services, whether offered in physical premises or online, within a broad definition of regulated fertility services would recognise the changing nature of wider fertility treatment.
- The Act should be amended to enable the removal of donor anonymity from the birth of any child born from donation. It was generally recognised by respondents to our consultation that the current system, where identifiable information is disclosed to the donor-conceived person at 18 and only on request, is being overtaken by other means (e.g. DNA consumer tests, social media) currently accessible to parents and donor-conceived individuals at an earlier age. We suggest that the Act is amended to allow the provision of identifiable information on donors after the birth of a child.
- Clinics should be required by law to inform donors and recipients of the potential for donor identity to be discovered through, for example, DNA testing websites, social media or further means other than the HFEA Register.
- The Act should require all donors and recipients to have implications counselling sessions before starting treatment to support informed consent and consideration of the long-term implications of the decision to provide or use donor gametes.
A summary of our proposals for change with reference to the Committee’s inquiry
Following public consultation the HFEA recommended changes to the HFE Act 1990 in November 2023.
The full proposals can be found on our website here, with those most applicable to egg donation or egg or embryo freezing set out below under the relevant theme.
Patient protection and safety
Patients should be assured that the treatment they are offered is safe, evidence-based and of a high standard. To provide this assurance, the HFEA wants to put patients at the heart of a revised law.
Fertility treatment is unique in modern healthcare – there is no comparable area of healthcare where a potential new life is created in a laboratory. But many of the regulatory issues we face are common to healthcare more generally - the Cumberlege report on women who suffered avoidable harm from private and NHS healthcare described a system that did not adequately recognise that patients are its primary purpose. Like all healthcare regulators, the HFEA needs to put patient protection and safety, alongside the long-established concern for the special status of the embryo, at the heart of its regulatory actions.
The proposals also recognised the significant changes in the fertility sector since the HFEA was set up. Today fertility treatment is provided predominantly through self-funding by patients, although this varies across the nations and regions of the UK. This is not to say that the interests of patients are not uppermost in the minds of clinic staff wherever patients are treated. However, the majority of clinics are now operating in a competitive market using advertising and other tools to attract new customers. Patients are having to make very difficult and expensive choices about medical care, in contrast to most medical interventions in the UK which can be accessed on the NHS. In addition, elements of fertility care and associated treatments are increasingly offered online or outside of our regulation.
In arguing for better patient protection and safety we do not wish to suggest that most clinics are putting patients at risk. They are not. Overall, the fertility sector has a good level of compliance with the law and our guidance, as our annual State of the Sector report demonstrates, with a low level of incidents per the number of cycles every year. But that does not diminish the need for appropriate regulatory tools where serious non-compliance occurs, however infrequently. At present the HFEA has no flexibility to adjust its powers according to regulatory performance. We want to reduce the regulatory burden for compliant clinics and focus our oversight on those that are not meeting the required regulatory standards.
The Act should be revised to include an over-arching focus on patient protection.
Patient protection should be an explicitly stated principle of the Act, with a requirement that HFEA decision-making and compliance by licensed clinics should have reference to it.
This proposal is intended to reflect the wider context of fertility treatment in the UK today, where the majority of patients pay for the treatment themselves (which is very unusual in UK healthcare) and consideration is not only about medical care and treatment, but as others have noted, about patients as consumers.
Our work with the Competition and Markets Authority and Advertising Standards Authority has emphasised the inadequacy of our regulatory powers in the modern market, and the CMA have recommended that this (and some other) aspects of the HFEA Code of Practice should be directly enforceable to help incorporate protection of patients’ consumer interests.
The Act should be revised to accommodate developments in the way fertility services are provided.
The HFEA is often approached by patients who feel we should be able to do more on a range of issues, from what is said on a clinic website, to the offer of treatment add-ons, to services offered outside of licensed clinics. The Act should be revised to ensure it remains relevant to today’s – and the developing – fertility market.
Bringing all related UK services, whether offered in physical premises or online, within a broad definition of regulated fertility services would recognise the changing nature of wider fertility treatment.
Access to donor information
When the Act was first introduced there was a general presumption that donation should be anonymous. Over time attitudes have changed, but the law does not fully reflect those changes. The current professional advice is that children benefit from learning from a young age that they have been conceived using donor gametes.
The issue of accessing donor information and identifying donors has become more urgent with the growing popularity of easily accessible, relatively affordable direct-to-consumer DNA testing and matching services, which have revolutionised our ability to find our genetic relatives, as well as the growth of social media groups to help individuals find their donor or donor siblings. Anonymity in relation to donor conception can therefore no longer be assured to donors or to donor-conceived people and to their genetic relatives.
Our recommendations in this section also respond to the fact that the decision to use donated gametes can have significant implications for the donor(s), the parent(s) and the donor-conceived individual(s). The decision might impact on existing and new personal relationships and family dynamics. Making sure that prospective parents have access to the right information on the use of donor gametes within their treatment is an important element of the treatment pathway to ensure that they can come to a properly informed decision.
The Act should be amended to enable the removal of donor anonymity from the birth of any child born from donation.
It was generally recognised by respondents to our consultation that the current system, where identifiable information is disclosed to the donor-conceived person at 18 and only on request, is being overtaken by other means (e.g. DNA consumer tests, social media) currently accessible to parents and donor-conceived individuals at an earlier age.
In recognition of this, we suggest that the Act is amended to allow the provision of identifiable information on donors after the birth of a child. This proposal is designed to ‘future proof’ the Act to recognise the increasing accessibility of information that parents are already able to find out about their donor should they choose to do so.
It is recognised that this is a significant departure from current practice and therefore a gradual approach to reach this position may be appropriate. Before any change to the law is implemented, there would need to be in-depth discussions with interested parties: professional bodies, patient and donor groups, donors and donor-conceived individuals, and licensed centres within the fertility sector.
Clinics should be required by law to inform donors and recipients of the potential for donor identity to be discovered through, for example, DNA testing websites, social media or further means other than the HFEA Register.
As part of the consent process, clinics should be legally required to inform donors and recipients about the possibility that any children born from donation could discover their donor’s identity before they are 18. This would change what is currently HFEA guidance into a legal requirement.
The Act should require all donors and recipients to have implications counselling sessions before starting treatment.
The complexity of donation and the potential impact that it can have over a lifetime on wider personal relationships makes it unusual among medical treatments. Therapeutic counselling must at its heart be a voluntary decision. However, there is a good case that the principle of properly informed consent requires all donors and recipients to have access to implications counselling sessions. This would allow the consideration of the long-term implications of their decision to provide or use donor gametes before starting treatment.
There have been some calls for further moves towards openness, such as making it compulsory to tell children if they are donor conceived (for example by adding such information to the birth registration certificate) and the retrospective removal of anonymity for those conceived from donation before the law was changed in 2005. The HFEA did not consider either of these suggestions in detail when it agreed its proposal for law reform.
Annex: HFEA data on egg donation and freezing
The data below is provided from the HFEA Register and covers various aspects of the inquiry Terms of Reference. Further data can be found on the HFEA dashboard. We have not provided data in relation to embryo donation as these will be included in the egg donation figures and relate to small numbers.
Data for 2020-2023 is preliminary and has not undergone validation. Data provided is from a live register and may not match data provided in previous requests or published elsewhere. HFEA Register data has a two year delay due to the need for the HFEA to receive outcome information from the treatments and validate, and process the data received from all UK licensed clinics.
Demographics of egg donors
The number of new egg donors slightly decreased from 1,495 in 2019 to 1,392 in 2023. The number of egg donors in the UK have remained around 1,500 per year since 2011 (Figure 1).
Figure 1. Number of new egg donors by year, 1991-2023
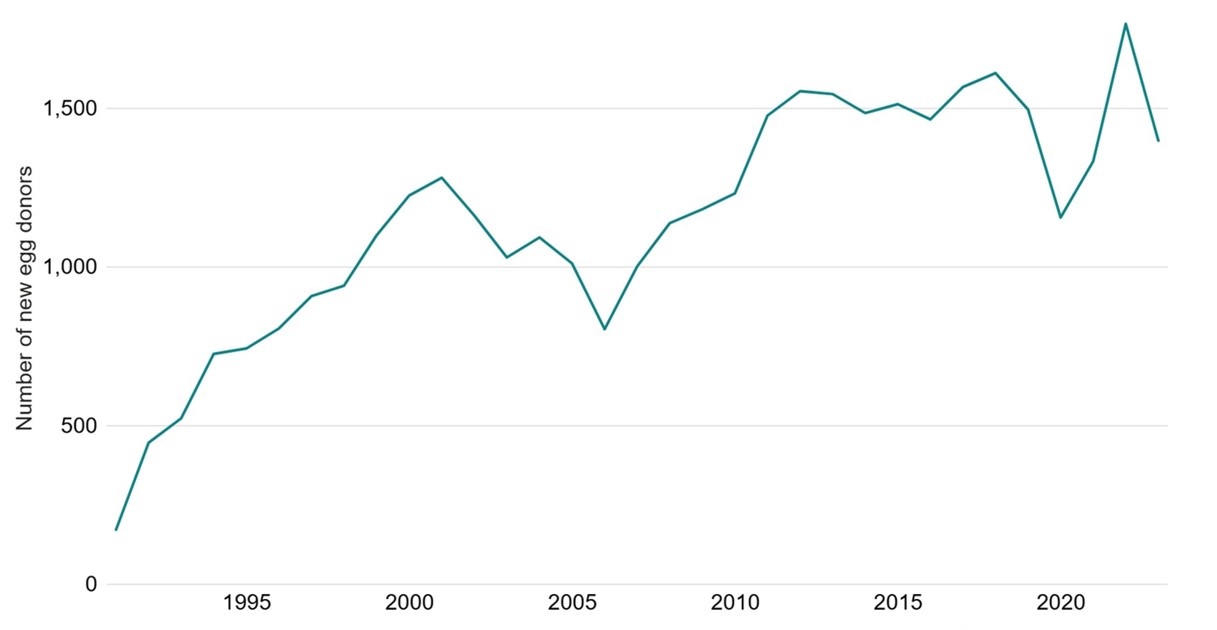
Note: Data from one clinic is excluded due to data reporting issues. This data uses new egg donor registrations only. Donors that registered multiple times will only be counted in the year of their first registration. All values have been rounded to the nearest 5. Numbers smaller than 8 have been suppressed as 0.
Source: HFEA dashboard
The median age of donors undergoing cycles to collect eggs for the primary reason of donation in 2019-2023 was 30, compared to a median age of 31 in 2009-2013.
| Table 1. Number of egg donors by year and donor age, 2009-2023 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Aged 18–24 | Aged 25–29 | Aged 30–35 | Aged 36–40 | Aged 41–45 | ||||||
| Year | Donors (% of total) | Avg. donation cycles | Donors (% of total) | Avg. donation cycles | Donors (% of total) | Avg. donation cycles | Donors (% of total) | Avg. donation cycles | Donors (% of total) | Avg. donation cycles |
| 2019-2023 | 1,463 (21%) | 1.3 | 2,005 (29%) | 1.37 | 3,182 (45%) | 1.36 | 568 (8%) | 1.16 | 102 (1%) | 1.19 |
| 2014-2018 | 1,154 (21%) | 1.32 | 1,669 (30%) | 1.39 | 2,477 (45%) | 1.37 | 442 (8%) | 1.14 | 53 (1%) | 1.21 |
| 2009-2013 | 445 (14%) | 1.19 | 808 (25%) | 1.18 | 1,594 (50%) | 1.21 | 375 (12%) | 1.13 | 31 (1%) | 1.16 |
| Note table 1: The percentages shown may not add up to 100% due to rounding and patients aging between different age bands within a 5-year period. One clinic has been excluded due to data reporting issues. | ||||||||||
Source: HFEA register
Previous HFEA research has found that egg donors lived in comparable areas to the general population (Figure 2). Donors were more commonly from deprived areas if they were younger, had overseas passports, or were from ethnic minority backgrounds.
Figure 2. Percentage of new egg donor registrations in England in each deprivation decile, 2011-2020
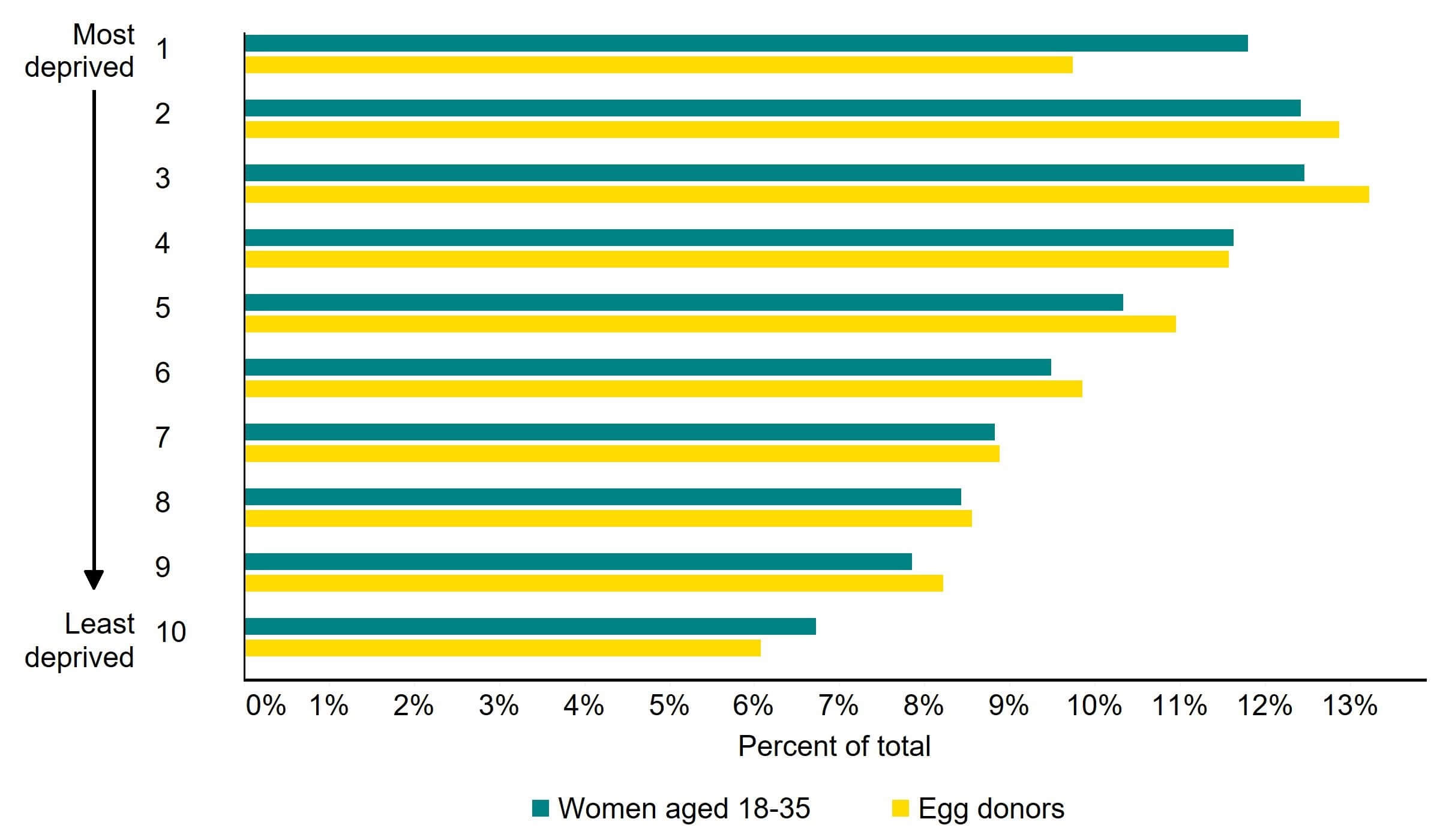
Note figure 2: This data includes egg donors aged 18-35 registering for the first time from 2011-2020 in England. Deprivation levels as in 2015 were used.
Egg donations used in fertility treatment in the UK are typically from UK donors, with only around 3% of donor eggs imported from abroad.17 One reason that patients may import donor sperm or eggs is to find a donor of matching ethnicity. Our 2021 National Patient Survey found that 82% of respondents said it was important that the ethnicity of a donor matched their own.
While most donor eggs used were from the UK, donations from donors of Other ethnicities were most likely to be imported (7%) followed by Black and Asian donors (3%).18
There are many more egg donors in the UK from White backgrounds than from Black, Asian or other ethnic groups. In 2023, there were around 55 new Black egg donors, 60 Asian, and 1,070 White.19
In 2020, around 1 in 3 (31%) of egg donors already had their own child.20
Data from the HFEA register shows that from 1991 to 2022, around 17,500 donors underwent cycles to collect eggs for the primary reason of donation.21 On average, these egg donors underwent 1.4 donation cycles, and fewer than 1% (0.7%) of these donors have undergone more than 5 egg donation cycles since 1991.
Demographics of patients freezing their eggs
Egg freezing cycles increased from around 2,600 in 2019 to nearly 7,000 in 2023 accounting for 7% of all cycles.22 Egg freezing cycles increased across all age groups from 2019 to 2023, with the largest increases among patients aged 30-37 (Figure 3). Egg freezing increased from 1,200 cycles in 2022 to 2,000 in 2023 (+67%) among patients aged 30-34 and from 1,500 to 2,300 (+53%) among patients aged 35-37. The average age of patients freezing their eggs was 35 and the average age of thawing was 40 in 2023.23
Figure 3. Number of egg freezing cycles by age group at freezing, 2019-23
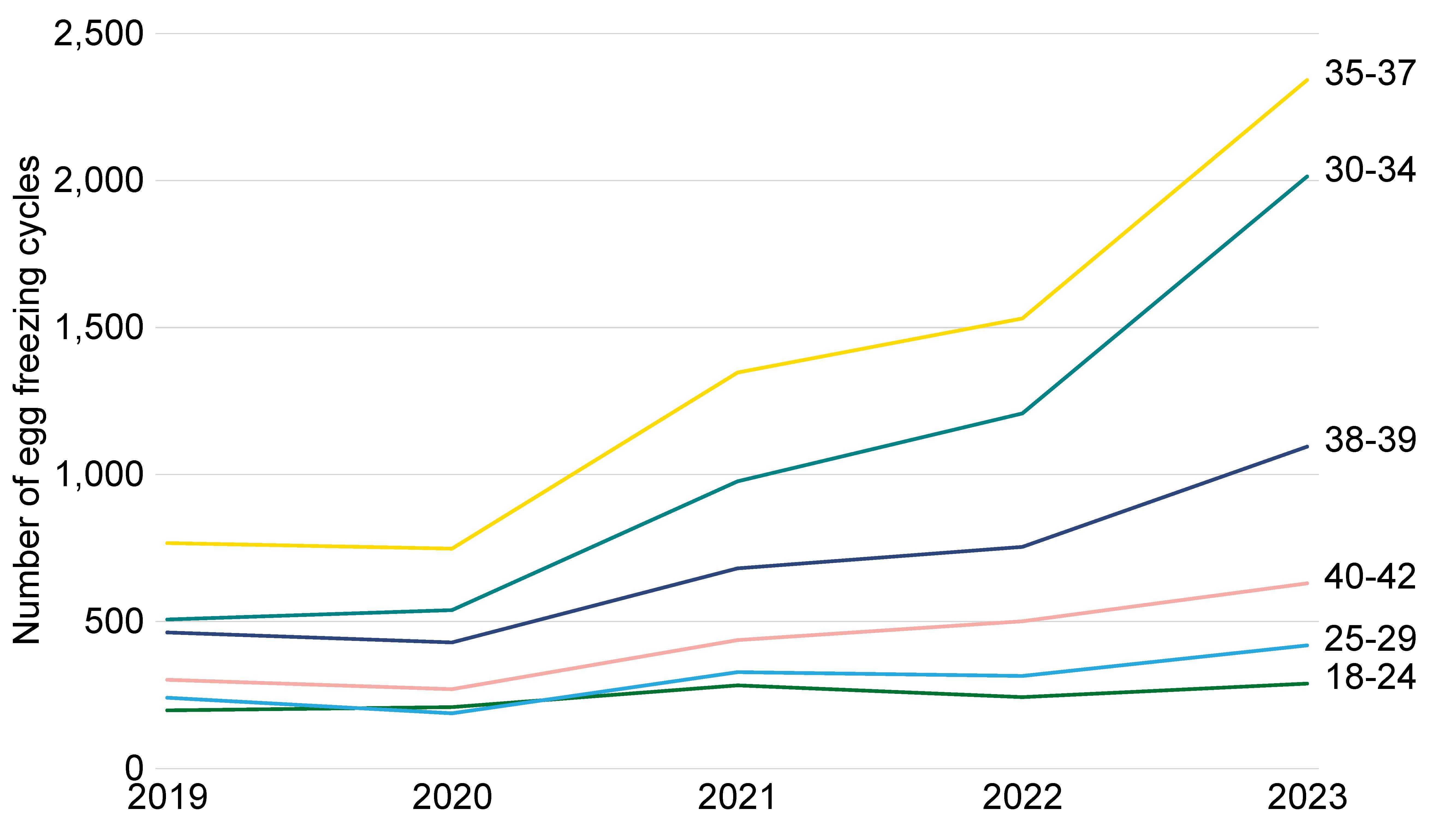
Note figure 3: Data excludes cycles where eggs/embryos were stored and donated in the same treatment.
Use of egg freezing was similar across ethnic groups in 2017-2021, but most used among patients of Other ethnic groups, with 5% of all cycles being egg freezing, followed by patients with a Black and a Mixed ethnic background (4%), and then White and Asian patients (3%).24
From 2018-2022, those storing eggs were mostly single patients, making up 89% of all egg freezing cycles.25 Opposite-sex couples accounted for 10% of egg freezing cycles, while female same-sex couples accounted for 1%. In contrast, the majority of those who thawed eggs for treatment were opposite-sex couples, accounting for 85% of egg thaw cycles. Single patients accounted for 13% of egg thawing cycles, while female same-sex couples accounted for 2%.
The HFEA does not collect data on the socioeconomic background of patients who freeze their eggs. Research is limited but suggests women undergoing elective egg freezing may be more likely to be from higher socioeconomic groups and have high levels of education.26
Motivations
Egg freezing data collected by the HFEA does not distinguish between reasons for freezing eggs, such as elective fertility preservation or for medical reasons. However, using NHS funding as a proxy for medical egg freezing indicates that at least 16% of egg freezing cycles were for medical reasons in 2018-22.27
Research on this topic is limited, but suggests that there are a range of reasons why patients may decide to freeze their eggs for non-medical reasons. The absence of the right partner with whom to start a family is often identified as a primary reason, alongside wanting to feel financially secure or ready for parenthood, and concerns about fertility declining with age.28 Social, cultural and religious approaches to motherhood and to assisted reproductive technologies have also been found to shape women’s perception of the procedure.29
Information and success rates
When looking at success rates for freezing eggs, numbers tend to be quite low. We advise patients to look at success rates for fresh IVF cycles with patients using their own eggs in their age band. We consider these rates to be more reliable as there are much higher numbers of fresh embryo transfers each year compared to egg freezing. This information can be accessed via our Choose a Fertility Clinic page or the HFEA dashboard.
Despite the large increase in the number of people freezing their eggs, our data shows the number of people returning to use their frozen eggs in treatment is much lower. There were nearly 7,000 egg freeze cycles in 2023, compared with 700 cycles with patients thawing their eggs.30
Egg freezing should not be presented as a guarantee to having a baby in the future, and we expect clinics to follow consumer law and advertising guidance for UK fertility clinics produced by the CMA and ASA.31
Number of individuals becoming eligible to request identifying information increases from 2023 onwards
Figure 4 shows the number of donor-conceived people who are eligible to access identifying information about their egg or sperm donor each year, on reaching the age of 18.
Figure 4. Donor-conceived people turning 18 born to identifiable donors, 2011-2037
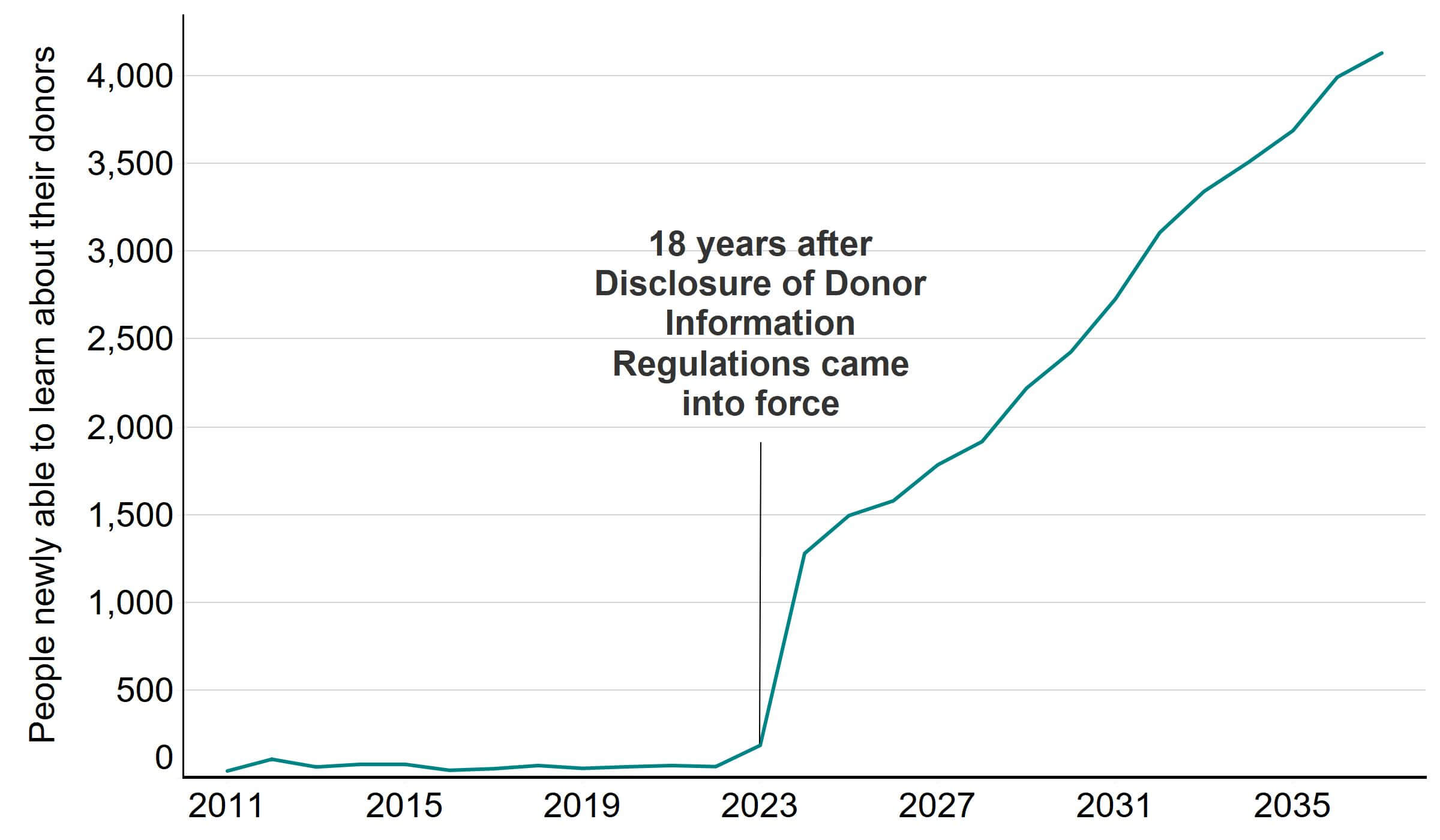
Note: This data includes all births as a result of IVF or DI in which an identifiable sperm or egg donor was used. Details of how donors were determined to be identifiable can be found in the Quality and methodology report.
Increasing number of applications for donation related information
The number of applications the HFEA receives for information about donation continues to grow and is expected to increase over the next few years as the number of children born from donation has risen. The number have risen particularly for female same-sex couples and single patients having treatment with donor sperm.
Figure 5. Average monthly Opening the Register applications that the HFEA has responded to
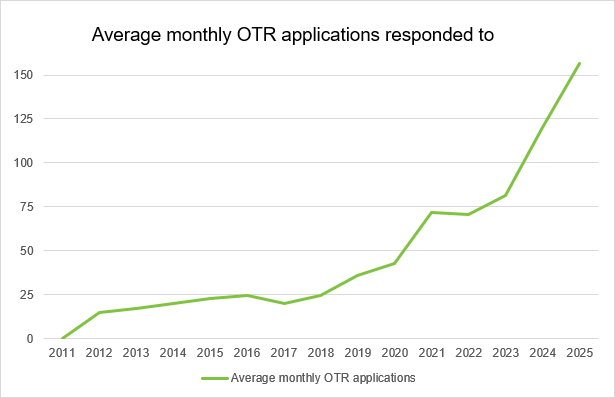
Source: HFEA
References
1Human Fertilisation and Embryology Authority (HFEA). The Fertility Sector 2024/25. 2025.
2HFEA Scientific and Clinical Advances Advisory Committee (SCAAC). from p67 of the papers, and from p8 of the minutes.
3HFEA Scientific and Clinical Advances Advisory Committee (SCAAC). Papers 9th June 2025. p79-81.
4Human Fertilisation and Embryology Authority (HFEA). National Patient Survey 2024. 2025.
5Human Fertilisation and Embryology Authority (HFEA). Trends in egg, sperm and embryo donation 2020. 2022.
6Human Fertilisation and Embryology Authority (HFEA). HFEA dashboard.
7Human Fertilisation and Embryology Authority (HFEA). HFEA Register data.
8Human Fertilisation and Embryology Authority (HFEA). Fertility treatment 2023: trends and figures. 2025.
9Human Fertilisation and Embryology Authority (HFEA). HFEA Register data.
10Human Fertilisation and Embryology Authority (HFEA). Egg donation: a factsheet.
11Human Fertilisation and Embryology Authority (HFEA). HFEA dashboard.
12Chehimi, M et al. Sociodemographic profile of women seeking elective egg freezing when financed by the public healthcare system. Reproductive BioMedicine Online. 2025.
13Human Fertilisation and Embryology Authority (HFEA). Family formations in fertility treatment 2022. 2024.
14Loyal, S. Reproductive timing and social egg freezing within British South Asian communities: a qualitative study. Journal of Reproductive and Infant Psychology. 2024; Jones BP, Kasaven L, L'Heveder A, et al. Perceptions, outcomes, and regret following social egg freezing in the UK; a cross-sectional survey. Acta Obstet Gynecol Scand. 2020.
15Loyal, S. Reproductive timing and social egg freezing within British South Asian communities: a qualitative study. Journal of Reproductive and Infant Psychology. 2024; Gürtin ZB, Morgan L, O'Rourke D, Wang J, Ahuja K. For whom the egg thaws: insights from an analysis of 10 years of frozen egg thaw data from two UK clinics, 2008-2017. J Assist Reprod Genet. 2019; Chehimi, M et al. Sociodemographic profile of women seeking elective egg freezing when financed by the public healthcare system. Reproductive BioMedicine Online. 2025.
16BBC News. Sperm donation: Judge warns over man who 'fathered 180 children'. 2025; BBC News. Sperm donor fathered 15 children despite DNA condition. 2022.
17Human Fertilisation and Embryology Authority (HFEA). Egg donation: a factsheet.
18Human Fertilisation and Embryology Authority (HFEA). Ethnic diversity in fertility treatment 2021. 2023.
19Human Fertilisation and Embryology Authority (HFEA). Egg donation: a factsheet.
20Human Fertilisation and Embryology Authority (HFEA). Egg donation: a factsheet.
21HFEA Scientific and Clinical Advances Advisory Committee (SCAAC). Minutes 9th June 2025. p9.
22Human Fertilisation and Embryology Authority (HFEA). Fertility treatment 2023: trends and figures. 2025.
23Human Fertilisation and Embryology Authority (HFEA). HFEA dashboard.
24Human Fertilisation and Embryology Authority (HFEA). Ethnic diversity in fertility treatment 2021. 2023.
25Human Fertilisation and Embryology Authority (HFEA). Family formations in fertility treatment 2022. 2024.
26Chehimi, M et al. Sociodemographic profile of women seeking elective egg freezing when financed by the public healthcare system. Reproductive BioMedicine Online. 2025.
27Human Fertilisation and Embryology Authority (HFEA). Family formations in fertility treatment 2022. 2024.
28Loyal, S. Reproductive timing and social egg freezing within British South Asian communities: a qualitative study. Journal of Reproductive and Infant Psychology. 2024; Jones BP, Kasaven L, L'Heveder A, et al. Perceptions, outcomes, and regret following social egg freezing in the UK; a cross-sectional survey. Acta Obstet Gynecol Scand. 2020.
29Loyal, S. Reproductive timing and social egg freezing within British South Asian communities: a qualitative study. Journal of Reproductive and Infant Psychology. 2024; Gürtin ZB, Morgan L, O'Rourke D, Wang J, Ahuja K. For whom the egg thaws: insights from an analysis of 10 years of frozen egg thaw data from two UK clinics, 2008-2017. J Assist Reprod Genet. 2019; Chehimi, M et al. Sociodemographic profile of women seeking elective egg freezing when financed by the public healthcare system. Reproductive BioMedicine Online. 2025.
30Human Fertilisation and Embryology Authority (HFEA). HFEA dashboard.
31Human Fertilisation and Embryology Authority (HFEA). HFEA backs new consumer law and advertising guidance for IVF clinics. 2021.
| Publication date: |
|---|

